Pharma Analysis Software
Pharma Analysis Software Specification
- Spare Parts
- Not applicable to software
- Features
- Automated report generation, data export to Excel/PDF, GMP/GLP compliance, audit trail management
- View Head
- Multiple display layouts
- Focus System
- Auto-focus compatible with digital imaging devices
- Theory
- Advanced analytical algorithms for pharmaceutical sample evaluation
- Drawtube
- Not applicable to software
- Sensor
- Compatible with various digital imaging devices
- Resolution
- Supports up to 1920 x 1080 pixels
- Interface
- User-friendly graphical interface with multi-user support
- Frame Rate
- Supports live imaging up to 60 FPS
- Focal Distance
- Not applicable to software
- Magnification
- Digital magnification options available, up to 400%
- Dimensions
- Digital software, no physical dimensions
- Focus Range
- Not applicable to software
- Eyepieces
- Not applicable to software
- Eyepiece Tube
- Not applicable to software
- Illumination
- Digital image enhancement features
- Coarse Adjustment Range
- Custom image contrast and brightness controls
- Fine Adjustment Range
- Advanced fine-tuning algorithms for detailed analysis
- Working Stage
- Not applicable to software
- Still Image Capture Resolution
- Up to 8 MP (depends on connected device)
- Video Capture Resolution
- 1080p full HD
- Image Format
- Supports JPG, PNG, TIFF, BMP formats
- Interpupillary Distance
- Not applicable to software
- Objective Achromatic
- Not applicable to software
- Condenser
- Not applicable to software
- Light Source
- Not applicable to software
- Data Backup
- Automated scheduled backups
- Third-Party Device Support
- Compatible with leading lab hardware brands
- User Manual
- Comprehensive digital manual provided
- Operating System Compatibility
- Windows 7/8/10/11, macOS, Linux
- Update Availability
- Regular online updates
- Statistical Tools
- Built-in statistics module for quality analysis
- Licensing
- Single-user and enterprise licensing options available
- Language Support
- Multi-language (English, Hindi, regional options)
- System Requirements
- Minimum 4GB RAM, 500MB disk space
- Security
- User authentication and encrypted data storage
- Database Integration
- Supports SQL and cloud-based data storage
- Customer Support
- 24x7 technical assistance via phone/email
Pharma Analysis Software Trade Information
- Minimum Order Quantity
- 1 Piece
- Delivery Time
- 3 Days
About Pharma Analysis Software
| Operating System | Window |
| Shape | Rectangular |
| Number of user | 01 |
| Language Support | English |
| USB Dongle | Yes |
Comprehensive Compatibility and Integration
The software runs smoothly across major operating systems (Windows, macOS, Linux) and integrates seamlessly with leading SQL and cloud database platforms. This ensures effortless data migration and scalability for diverse laboratories while offering compatibility with a wide range of lab imaging devices and hardware.
Secure and Reliable Data Management
User authentication, encrypted data storage, and automated scheduled backups guarantee that sensitive pharmaceutical data is protected at all times. Compliance features such as audit trail management and GMP/GLP alignment provide confidence for regulatory environments and easy tracking of all data modifications.
Intuitive Interface and Advanced Analysis
Featuring a modern, multi-user graphical interface, the software offers digital magnification up to 400%, live imaging up to 60 FPS, and support for popular image formats. Powerful statistics tools and fine-tuning algorithms enhance the quality of analysis, while automated report generation simplifies sharing and compliance.
FAQ's of Pharma Analysis Software:
Q: How does the software ensure data security and confidentiality?
A: Pharma Analysis Software offers robust security through user authentication and encrypted data storage. Automated backups further ensure that data is safe from loss, and compliance with GMP/GLP standards safeguards sensitive information for regulatory and auditing needs.Q: What are the operating system and hardware requirements for installation?
A: The software is compatible with Windows 7/8/10/11, macOS, and Linux. Minimum hardware requirements include 4GB RAM and 500MB of disk space, ensuring efficient performance on modern computers.Q: How can I integrate the software with lab hardware and databases?
A: It supports seamless integration with major SQL and cloud databases. Additionally, the software is compatible with leading laboratory imaging devices and hardware brands, allowing for smooth connection and data flow in any lab environment.Q: What resources are available to help users learn and operate the software?
A: A comprehensive digital user manual is provided, detailing all features and functionalities. Additionally, users have access to 24x7 technical support via phone and email to resolve any queries or issues.Q: When are software updates available?
A: Regular updates are provided online, keeping the software up to date with the latest analytical algorithms, security features, and compatibility improvements.Q: What benefits does the software offer for pharmaceutical analysis and compliance?
A: It includes built-in statistics modules, automated report generation, and audit trail management, making it easier to meet GMP/GLP compliance and produce reliable, audit-ready results. The software's advanced algorithms and high-resolution imaging facilitate accurate and detailed sample evaluation.Q: Is the software available in multiple licensing and language options?
A: Yes, it offers both single-user and enterprise licensing models and supports multiple languages, including English, Hindi, and other regional languages, catering to diverse user needs across India and beyond.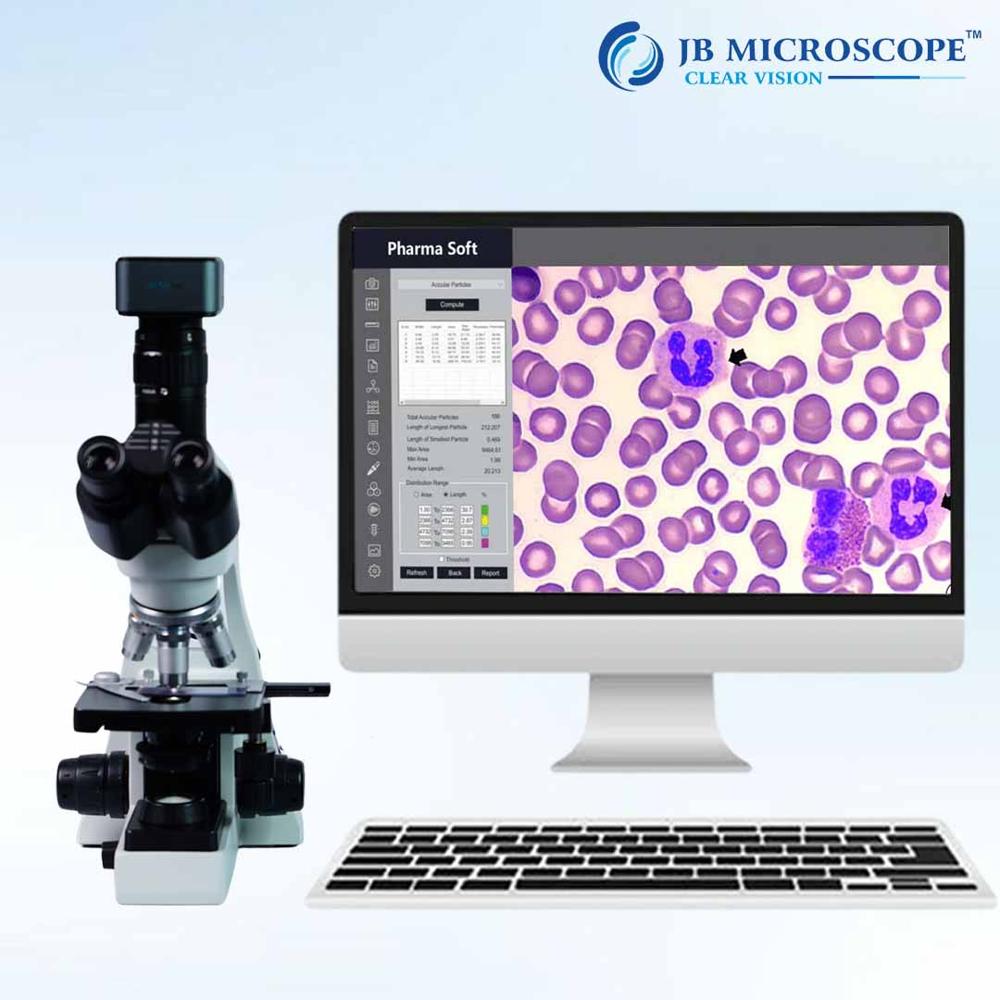
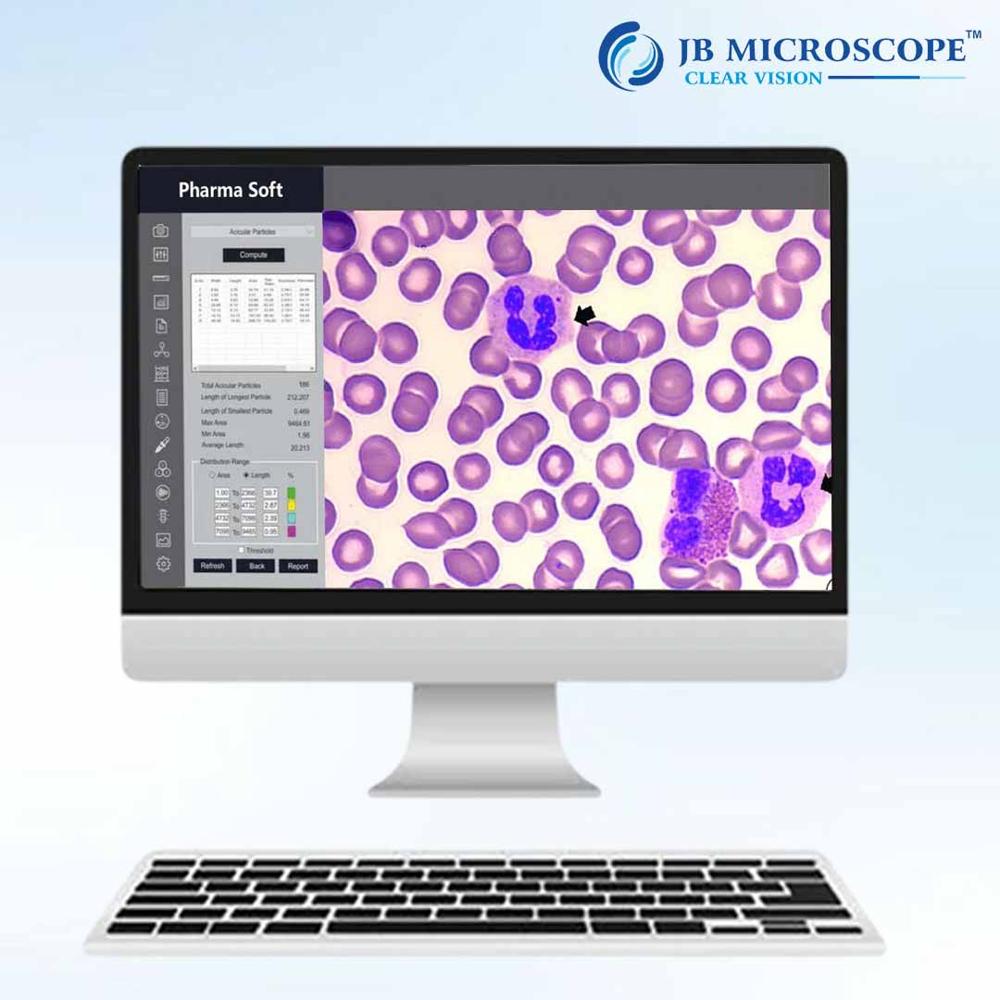
Price 35200.0 INR/ Piece
- Minimum Order Quantity
- 1 Piece
- Delivery Time
- 3 Days

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Software Category
Metallurgical Microscope Software
Price 28000.0 INR / Piece
Minimum Order Quantity : 1 Piece
Resolution : Up to 5MP (2592 x 1944 pixels)
Video Capture Resolution : 1280 x 720 pixels
Objective Achromatic : Plan Achromatic 4X, 10X, 40X, 100X (oil)
Magnification : 40X to 1600X (with eyepieces and objectives)
Textile Analysis Software
Price 41000.0 INR / Piece
Minimum Order Quantity : 1 Piece
Resolution : 1920 x 1080 pixels (Full HD)
Video Capture Resolution : 1920 x 1080 pixels at 30/60 fps
Objective Achromatic : 4x, 10x, 40x, 100x objectives included
Magnification : Up to 1000x with digital zoom
Weld Check Measurement Software
Price 32500.0 INR / Piece
Minimum Order Quantity : 1 Piece
Resolution : Up to 1200 DPI (software dependent on camera hardware)
Video Capture Resolution : Full HD 1920x1080
Objective Achromatic : Yes, for optical distortion elimination
Magnification : 1X to 200X (combined digital and optical)
Particle Size Software
Price 32000.0 INR / Piece
Minimum Order Quantity : 1 Piece
Resolution : 1920 x 1080 pixels
Video Capture Resolution : 1080p FHD
Objective Achromatic : 4x, 10x, 40x (S), 100x (oil)
Magnification : 40x1000x (objectivedependent)